Translate this page into:
Extracorporeal Membrane Oxygenation in Cardiogenic shock – A Review
*Corresponding author: Yatin Mehta, Department of Critical Care and Anestehsiology, Medanta Institute of Critical Care and Anesthesiology, Medanta - The Medicity, Gurugram, Haryana, India. yatinmehta@hotmail.com
-
Received: ,
Accepted: ,
How to cite this article: Nambiar PM, Sharma J, Mehta Y. Extracorporeal membrane oxygenation in cardiogenic shock – A review. J Card Crit Care TSS 2023;7:35-9.
Abstract
Extracorporeal membrane oxygenation has been used in adult in cardiogenic shock due to myocardial infarction, myocardiopathy, myocarditis, or inability to come off cardiopulmonary bypass (CPB) after cardiac surgery.3 ECMO can provide partial or total support, but it is temporary. It is not a definitive treatment but a life support system that allows time for evaluation, diagnosis and treatment of the condition that causes heart failure. With high incidence of late stage coronary artery disease and infective myocarditis in India, the usage of ECMO for refractory cardiogenic shock is going to be a promising approach for effective management of refractory heat failure .ECMO has proven to be an effective therapeutic modality in managing cardiovascular collapse as a bridge to recovery or more definitive treatment options.
Keywords
VA ECMO
cardiogenic shock
complications
contraindications on ECMO
INTRODUCTION
There are various modalities to manage cardiogenic shock. Mechanical support to heart and lung is still considered to be mainstay of the treatment. When there is evidence of inadequate tissue oxygen delivery despite intra-aortic balloon counterpulsation, invasive ventilation, and inotropes, full circulatory support is found to have a significant impact on the patient outcome. Extracorporeal membrane oxygenation (ECMO) can subsume the function of both heart and lungs.
ECMO was first used successfully for life-threatening respiratory failure in an adult in 1971[1] and in a neonate with meconium aspiration in 1975.[2] ECMO has been used in adult in cardiogenic shock due to myocardial infarction, myocardiopathy, myocarditis, or inability to come off cardiopulmonary bypass after cardiac surgery[3] ECMO can provide partial or total support, but it is temporary. It is not a definitive treatment but a life support system that allows time for evaluation, diagnosis, and treatment of the condition that causes heart failure [Figure 1].
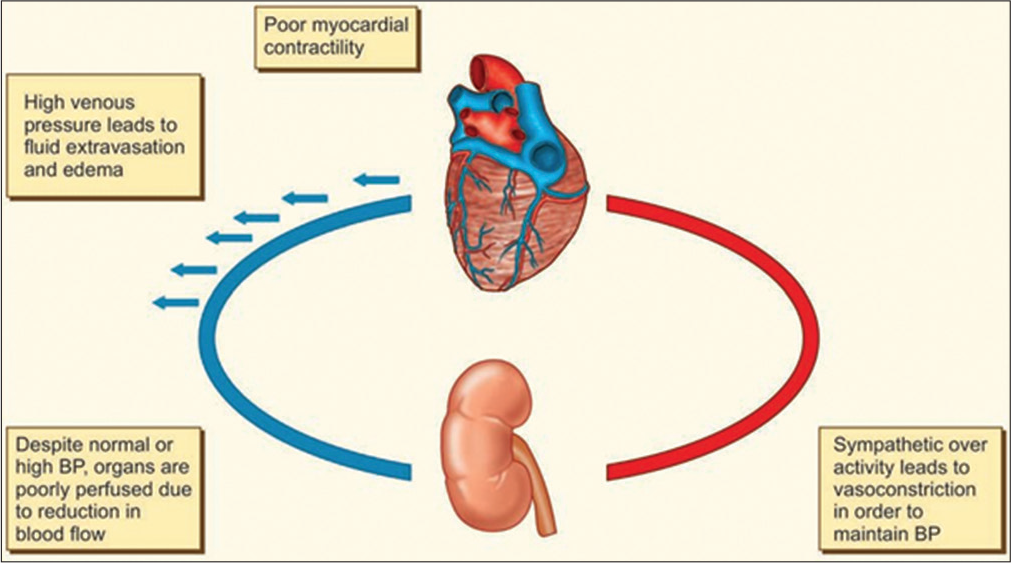
- Cardiogenic shock-sequence of events.
This modality requires a high level of care and traditionally has remained underutilized. ECMO team should consider the patient condition, prognosis of the disease, and weaning strategies before embarking on the task of putting an ECMO as it has lot of implications.
CARDIOGENIC SHOCK AND ECMO
ECMO controls gas exchange and perfusion, stabilizes the patient physiologically, decreases the risk of on-going iatrogenic injury, and allows ample time for diagnosis, treatment, and recovery from primary injury or disease of the heart.
It can be broadly categorized into two types:
Venovenous ECMO (VV-ECMO)
Venoarterial ECMO (VA-ECMO).
VV-ECMO is appropriate only for respiratory failure.
VA-ECMO is used for cardiogenic shock and is currently the fastest growing indication for ECMO worldwide.[4]
In ECMO, the right atrial venous blood is drained through a large cannula, pumped through an artificial lung (oxygenator), and back into the patient either into the aorta (venoarterial and VA mode) or into right atrium (venovenous, VV mode). Venoarterial access puts the oxygenator in parallel with native lung and substitutes for both heart and lung functions. Venovenous access puts at artificial lung in series with the native lung.
In adults, blood is usually removed through a femoral vein and returned through a femoral artery (peripheral ECMO). Occasionally, other cannulation strategies, such as directly cannulating the right atrium and aorta (central ECMO), may be employed. For total support, the blood flow required is 60–100 mL/kg/min (equal to cardiac output) [Figure 2]. The target MVO2 is 60–80% and avoid full flows as far as possible [Tables 1 and 2].
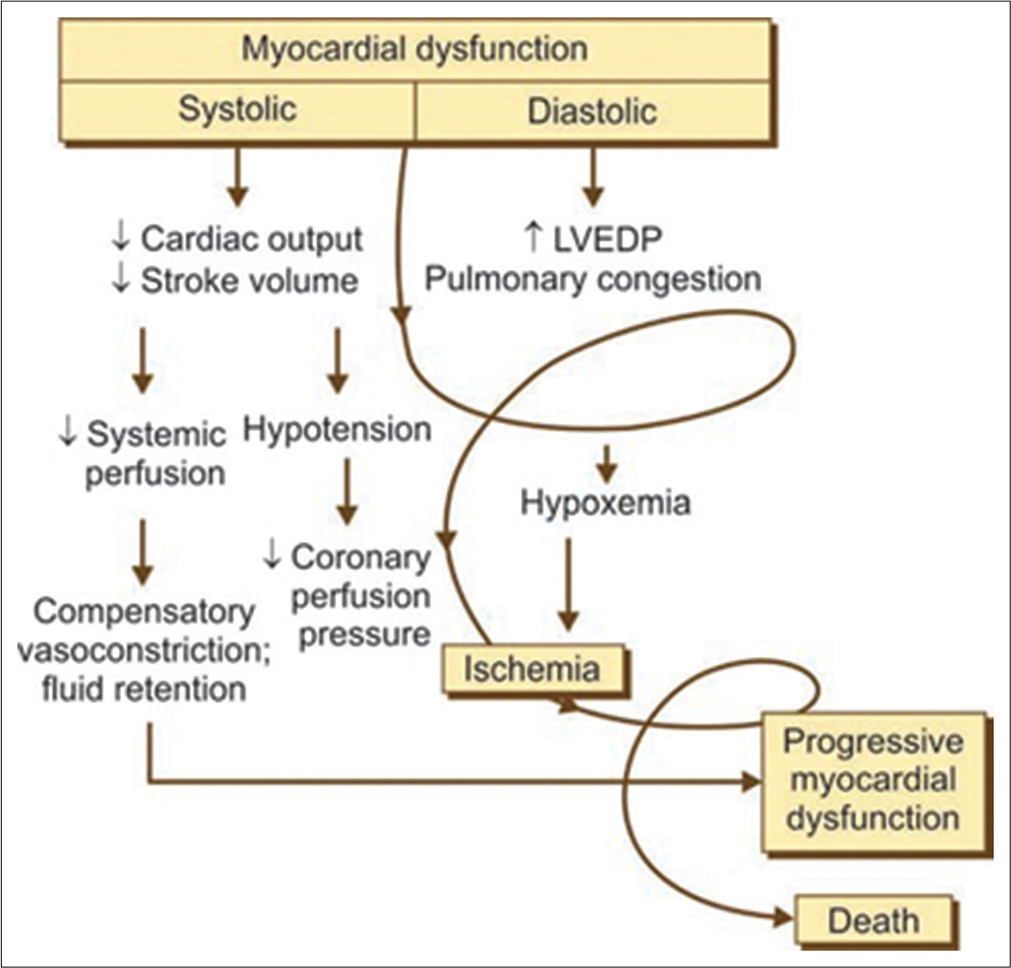
- The Downfall Spiral.
| • ScVO2 >70% • MAP >60 mm Hg (in case of full support with nonpulsatile flow) • MAP >70 mm Hg (in case of partial support with pulsatile flow) • Avoid full flow and give partial flow about 2/3 through ECMO • Maintain oxygen saturation of 90% or PaO2 of >50 mm Hg (in young patient) and >60 (in elderly and with CAD or CVA) • Maintain coronary saturation of >70% |
ScVO2: Mixed venous Oxygen saturation, MAP: Mean arterial pressure, CAD: Coronary artery disease, CVA: Cerebrovascular accident, ECMO: Extracorporeal membrane oxygenation
| • Flow: 60–80 cc/kg/min • FiO2: 100% • MvO2: 60–75% • SpO2: 95–100% • pCO2: 35–45 mm Hg • MAP: 60–90 mm Hg • pH: 7.35–7.45 • Platelet count: >80,000 • Hematocrit: >28% |
MAP: Mean arterial pressure
Peripheral ECMO is less invasive, is easier to place, and can be placed percutaneously by surgeons or intensivists. It can be initiated quickly, making it more appropriate in emergencies. However, the cardiac output of the failing heart competes with retrograde ECMO flow from the femoral aortic cannula, producing admixing in the thoracic aorta and an increase in left ventricular wall tension. If there is concomitant respiratory failure, this can result in the delivery of inadequately oxygenated blood to the coronary and cerebral circulations and hinder recovery.[5]
Central ECMO is not associated with this problem but is slower to initiate and may have a higher complication rate with bleeding and infection. It is usually confined to the support of patients after surgical revascularization and is initiated often in the operating room.
Cardiopulmonary resuscitation along with ECMO (E-CPR) is yet another emerging use of VA-ECMO in restoring the circulation during cardiac arrest when used in conjunction with advanced life support strategies. Data show improved in hospital survival with fairly good neurological outcomes for up to 2 years when VA-ECMO is used along with CPR in highly selected patients.[6] As per a systematic review and meta-analysis done by Wilson-Smith et al. the survival rates in VA-ECMO for refractory cariogenic shock at 1–5 years were 36.7%, 33.8%, 31.7%, and 29.9%, respectively.[7]
Peripheral VA-ECMO is adequate for most forms of cardiogenic shock, but frequent echocardiography is necessary to monitor for progressive ventricular dilatation and, increased left ventricular end diastolic pressure which can put strain on the left venticle, leading to pulmonary edema and increase in myocardial oxygen consumption. If this develops, the left atrium can be vented either by changing the ECMO circuit configuration or by performing a percutaneous atrial septostomy. Use of LEFT ventricular assist devices, Impella, also has proven to be an effective strategy to unload such ventricle.[8-11]
| • Irreversible disease (e.g., malignancy) • Dilated cardiomyopathy except for waiting for final destination therapy or transplant • Age >75 years • Patient on ventilator from 15 days • Intracranial bleed • Active bleeding from non-compressive site • Patient of gross multiorgan failure • Severe CNS injury including encephalitis, persistent vegetative state, hypoxic ischemic encephalopathy, and intractable seizures • Unwitnessed arrest or arrest >30 min |
ECMO: Extracorporeal membrane oxygenation
| • Severe aortic regurgitation • Severe aortic stenosis • Coarctation of aorta • Aortic dissection/Aneursym |
The clinical and echocardiographic contraindications are as mentioned in [Tables 3 and 4]. In septic patient, initiation of ECMO is controversial even in the presence of cardiogenic shock due to presence of conduit bacteremia, inflammation, and bleeding diathesis in septic patients with disseminated intravascular coagulopathy. However, advancement in extracorporeal technology and clinical practice might minimize these complications and that potential benefits may outweigh the risks. A prospective trial found that preexisting sepsis is not a contraindication for ECMO; however, in septic patient, VA ECMO should be used with caution [Tables 5 and 6].[12]
| Cardiogenic shock secondary to acute coronary syndrome |
|---|
| • Viral myocarditis • Non ischemic cardiomyopathy including sepsis induced cardiomyopathy • Cardiogenic shock with acute severe mitral regurgitation and aortic regurgitation • Pulmonary hypertension and right heart failure • Pulmonary embolus with hemodynamic collapse • Cardiac arrest (e-CPR) • Postcardiotomy cardiogenic shock • Failure to wean off from cardiopulmonary bypass • Poisoning—beta blocker, calcium channel blocker, aluminum phosphide • Patient waiting for left ventricular assist device • Patient waiting for cardiac transplant |
ECMO: Extracorporeal membrane oxygenation
| Presence of any two criteria mentioned below observed over a period of 4–6 h after maximum medical resuscitation: • Refractory arrhythmias • Cardiogenic shock with high inotropic requirements • Lactate level >5 mmoL/L or rising titer or ScVO2 <65%with maximum medical management • Signs of poor tissue perfusion |
ECMO: Extracorporeal membrane oxygenation
European Society of Cardiology guidelines for the diagnosis and treatment of acute and chronic heart failure recommend short-term mechanical circulatory support (as a “bridge to recovery”) in patients remaining severely hypoperfused despite inotropic therapy and with a potentially reversible cause (e.g., viral myocarditis) or a potentially surgically correctible cause (acute inter ventricular septal rupture).[13]
COMPLICATIONS
Bleeding
VA ECMO is associated with bleeding in 30–60% of cases,[13,14] sometimes requiring massive transfusions. New pumps and improved circuit biocompatibility allow lower levels of anticoagulation to be used and should reduce the impact of this complication.
Neurological events
Clotting abnormalities predispose to hemorrhagic stroke, which, combined with circuit embolic complications such as air bubbles or clots, results in an overall stroke rate of 3–12%.[4,14,15]
Nosocomial infection
Mechanical complications
Device and circuit complications appear to be declining.[4]
Disastrous complications when on ECMO for cardiogenic shock
Limb ischemia – a known complication of VA-ECMO, cannula size and position playing a major deciding factor. Use of a reperfusion catheter to perfuse the distal extremity beyond the entry site of ECMO cannula increases the likelihood of limb preservation. This is commonly done with a catheter based insertion of a reperfusion cannula through retrograde insertion from distal limb vessel or with an end to side graft from the ECMO circuit into the superficial femoral artery.[17]
Other complications which may be seen are hemolysis, hepatic dysfunction, renal dysfunction, hypertension, and device failure (clot, oxygenator, cannula and pump malfunction, and cracks in circuit).
With high incidence of late stage coronary artery disease and infective myocarditis in India, the usage of ECMO for refractory cardiogenic shock is going to be a promising approach for effective management of refractory heart failure.[18]
CONCLUSION
ECMO has proven to be an effective therapeutic modality in managing cardiovascular collapse as a bridge to recovery or more definitive treatment options. Early initiation is the key to a successful outcome in cardiogenic shock. A well-coordinated ECMO team is the core factor of management of ECMO. As with any technology used in critically ill patients, end of life discussion and target of therapy and prognosis are essential. Daily update to the family members of the patient with regard to the patient’s condition is crucial due to the financial burden associated with it in the modern health-care scenario. Palliative care consultations when available also should be initiated early.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Prolonged extracorporeal oxygenation for acute post-traumatic respiratory failure (shock-lung syndrome) Use of the Bramson membrane lung. N Engl J Med. 1972;286:629-34.
- [CrossRef] [PubMed] [Google Scholar]
- Extracorporeal membrane oxygenation (ECMO) cardiopulmonary support in infancy. Trans Am Soc Artif Intern Organs. 1976;22:80-93.
- [Google Scholar]
- Extracorporeal membrane oxygenation to support adult patients with cardiac failure: Predictive factors of 30-day mortality. Interact Cardiovasc Thorac Surg. 2010;10:721-6.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of extracorporeal membrane oxygenation on coronary perfusion and regional blood flow distribution. Pediatr Res. 1992;31:80-4.
- [CrossRef] [PubMed] [Google Scholar]
- Two-year survival and neurological outcome of in-hospital cardiac arrest patients rescued by extracorporeal cardiopulmonary resuscitation. Int J Cardiol. 2013;168:3424-30.
- [CrossRef] [PubMed] [Google Scholar]
- Outcomes of venoarterial extracorporeal membrane oxygenation for refractory cardiogenic shock: Systematic review and meta-analysis. Ann Cardiothorac Surg. 2019;8:1-8.
- [CrossRef] [PubMed] [Google Scholar]
- Balloon atrial septostomy for left ventricular decompression in patients receiving extracorporeal membrane oxygenation for myocardial failure. J Pediatr. 1993;122:S95-9.
- [CrossRef] [PubMed] [Google Scholar]
- Decompression of the left atrium during extracorporeal membrane oxygenation using a transseptal cannula incorporated into the circuit. Crit Care Med. 2006;34:2603-6.
- [CrossRef] [PubMed] [Google Scholar]
- Left heart decompression by atrial stenting during extracorporeal membrane oxygenation. Int J Artif Organs. 2009;32:240-2.
- [CrossRef] [PubMed] [Google Scholar]
- Should ECMO be used in cardiogenic shock? Crit Care. 2019;23:174.
- [CrossRef] [PubMed] [Google Scholar]
- Survival of septic adults compared with nonseptic adults receiving extracorporeal membrane oxygenation for cardiopulmonary failure: A propensity-matched analysis. J Crit Care. 2013;28:532-e1.10.
- [CrossRef] [PubMed] [Google Scholar]
- ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The task force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European society of cardiology. Developed in collaboration with the heart failure association (HFA) of the ESC. Eur Heart J. 2012;33:1787-847.
- [Google Scholar]
- Outcomes and long-term quality-of-life of patients supported by extracorporeal membrane oxygenation for refractory cardiogenic shock. Crit Care Med. 2008;36:1404-11.
- [CrossRef] [PubMed] [Google Scholar]
- Extracorporeal membrane oxygenation with a polymethylpentene oxygenator (Quadrox D). The experience of a single Italian centre in adult patients with refractory cardiogenic shock. ASAIO J. 2008;54:89-94.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical experience with 202 adults receiving extracorporeal membrane oxygenation for cardiac failure: Survival at five years. J Thorac Cardiovasc Surg. 2001;122:92-102.
- [CrossRef] [PubMed] [Google Scholar]
- Side-arm grafts for femoral extracorporeal membrane oxygenation cannulation. Ann Thorac Surg. 2012;94:e111-2.
- [CrossRef] [PubMed] [Google Scholar]
- Use of extracorporeal membrane oxygenation in adults. Heart Lung Circ. 2014;23:10-23.
- [CrossRef] [PubMed] [Google Scholar]






